COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D07KVV
|
|||
| Drug Name |
Selinexor
|
|||
| Synonyms |
Xpovio; KPT 330; KPT-330; KPT-330(Selinexor); KPT330;Selinexor; 1393477-72-9; 31TZ62FO8F; CHEMBL3545185; SCHEMBL14678327; Selinexor; Selinexor (KPT-330); Selinexor [USAN:INN]; Tube706; UNII-31TZ62FO8F
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Coronavirus Disease 2019 (COVID-19) | Phase 2 | [1] | |
| Other Indication | Multiple myeloma | Approved | [2] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Company |
Karyopharm therapeutics
|
|||
| Structure |
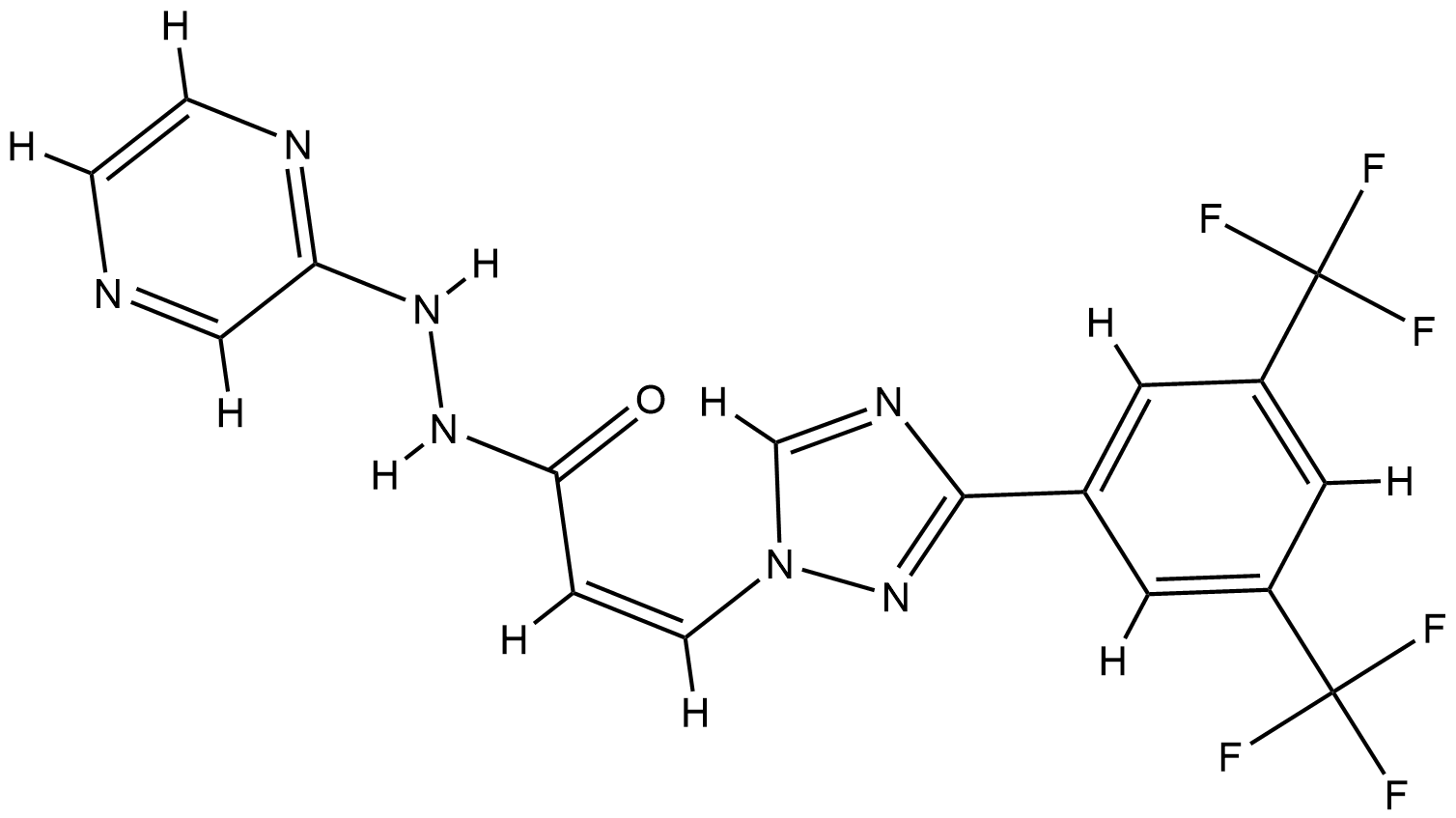 |
Download2D MOL |
||
| Formula |
C17H11F6N7O
|
|||
| Canonical SMILES |
C1=CN=C(C=N1)NNC(=O)C=CN2C=NC(=N2)C3=CC(=CC(=C3)C(F)(F)F)C(F)(F)F
|
|||
| InChI |
1S/C17H11F6N7O/c18-16(19,20)11-5-10(6-12(7-11)17(21,22)23)15-26-9-30(29-15)4-1-14(31)28-27-13-8-24-2-3-25-13/h1-9H,(H,25,27)(H,28,31)/b4-1-
|
|||
| InChIKey |
DEVSOMFAQLZNKR-RJRFIUFISA-N
|
|||
| CAS Number |
CAS 1393477-72-9
|
|||
| PubChem Compound ID | ||||
| PubChem Substance ID | ||||
| References | Top | |||
|---|---|---|---|---|
| 1 | ClinicalTrials.gov (NCT04349098) Evaluation of Activity and Safety of Oral Selinexor in Participants With Severe COVID-19 Infection. U.S. National Institutes of Health. | |||
| 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

