COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D07WQT
|
|||
| Drug Name |
Ivermectin
|
|||
| Synonyms |
IVERMECTIN; Ivermectin B1a; Dihydroavermectin B1a; 70288-86-7; 22,23-Dihydroavermectin B1a; avermectin H2B1a; UNII-91Y2202OUW; 71827-03-7; CHEBI:63941; 91Y2202OUW; Ivermectin
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Coronavirus Disease 2019 (COVID-19) | Phase 2/3 | [1] | |
| Other Indication | Onchocerciasis | Approved | [2] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Structure |
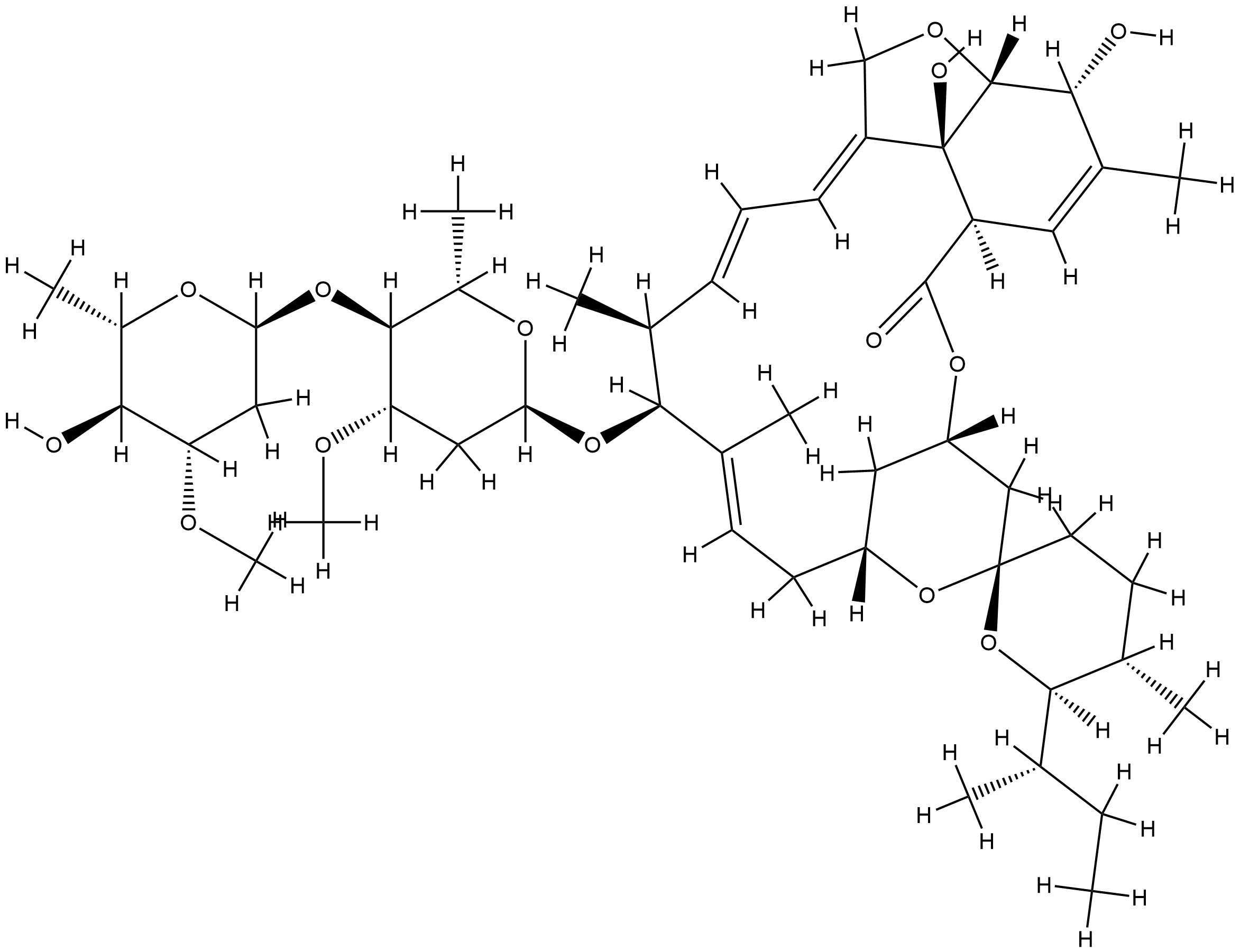 |
Download2D MOL
|
||
| Formula |
C48H74O14
|
|||
| Canonical SMILES |
CCC(C)C1C(CCC2(O1)CC3CC(O2)CC=C(C(C(C=CC=C4COC5C4(C(C=C(C5O)C)C(=O)O3)O)C)OC6CC(C(C(O6)C)OC7CC(C(C(O7)C)O)OC)OC)C)C
|
|||
| InChI |
1S/C48H74O14/c1-11-25(2)43-28(5)17-18-47(62-43)23-34-20-33(61-47)16-15-27(4)42(26(3)13-12-14-32-24-55-45-40(49)29(6)19-35(46(51)58-34)48(32,45)52)59-39-22-37(54-10)44(31(8)57-39)60-38-21-36(53-9)41(50)30(7)56-38/h12-15,19,25-26,28,30-31,33-45,49-50,52H,11,16-18,20-24H2,1-10H3/b13-12+,27-15+,32-14+/t25-,26-,28-,30-,31-,33+,34-,35-,36-,37-,38-,39-,40+,41-,42-,43+,44-,45+,47+,48+/m0/s1
|
|||
| InChIKey |
AZSNMRSAGSSBNP-XPNPUAGNSA-N
|
|||
| CAS Number |
CAS 71827-03-7
|
|||
| PubChem Compound ID | ||||
| ChEBI ID |
CHEBI:63941
|
|||
| References | Top | |||
|---|---|---|---|---|
| 1 | ClinicalTrials.gov (NCT04351347) The Efficacy of Ivermectin and Nitazoxanide in COVID-19 Treatment. U.S. National Institutes of Health. | |||
| 2 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 202736 | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

