Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D0Y8UV
|
|||
| Former ID |
DND000015
|
|||
| Drug Name |
EMD-1204831
|
|||
| Indication | Solid tumour/cancer [ICD-11: 2A00-2F9Z; ICD-10: C00-D48; ICD-9: 140-199, 210-229] | Phase 1 | [1] | |
| Company |
Merck KGaA
|
|||
| Structure |
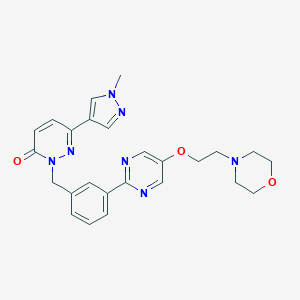 |
Download2D MOL |
||
| Formula |
C25H27N7O3
|
|||
| Canonical SMILES |
CN1C=C(C=N1)C2=NN(C(=O)C=C2)CC3=CC(=CC=C3)C4=NC=C(C=N4)OCCN5CCOCC5
|
|||
| InChI |
1S/C25H27N7O3/c1-30-18-21(14-28-30)23-5-6-24(33)32(29-23)17-19-3-2-4-20(13-19)25-26-15-22(16-27-25)35-12-9-31-7-10-34-11-8-31/h2-6,13-16,18H,7-12,17H2,1H3
|
|||
| InChIKey |
CIUKPBWULKEZMF-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 1100598-15-9
|
|||
| PubChem Compound ID | ||||
| References | Top | |||
|---|---|---|---|---|
| REF 1 | ClinicalTrials.gov (NCT01110083) First-in-Man, Dose-escalation Trial of c-Met Kinase Inhibitor EMD 1204831 in Subjects With Advanced Solid Tumors. U.S. National Institutes of Health. | |||
| REF 2 | 2012 ASCO Annual Meeting. | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

