Prodrug Information
| Prodrug General Information | Top | |||||
|---|---|---|---|---|---|---|
| Prodrug ID |
DCXV01
|
|||||
| Prodrug Name |
Debio 1450
|
|||||
| Synonyms |
Afabicin; UNII-DMM8663H2R; DMM8663H2R; 1518800-35-5; Afabicin [INN]; Debio1450; Debio 1450; CHEMBL4297501; SCHEMBL15479062; SCHEMBL16262317; GTPL10754; DB15268; HY-109000; CS-0030430; Q27276486;
Click to Show/Hide
|
|||||
| Indication | Skin and skin-structure infection [ICD-11: 1F28-1G0Z] | Phase 2 | [1] | |||
| Activation |
Prodrug 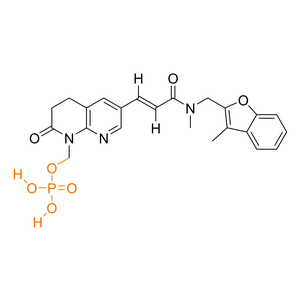
|

|
Parent Drug 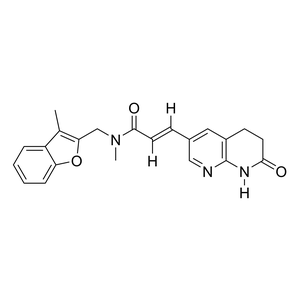
|
|||
| 2D MOL 3D MOL | 2D MOL 3D MOL | |||||
|
(1) Bioconversion Enzyme:
Alkaline phosphatase
(EC 3.1)
|
[2] | |||||
| Prodrug Strategy |
Classical prodrug strategy
[Carrier linked prodrug]
|
|||||
| Improved property |
Increase solubility; Increase oral absorption
|
[3] | ||||
| Description |
Debio 1452 is a BCS Class II drug with limited aqueous solubility and oral bioavailability, whereas its prodrug Debio 1450 has a dramatically improved oral bioavailability and solubility profile.
|
[3] | ||||
| Formula |
C23H24N3O7P
|
|||||
| Canonical SMILES |
CC1=C(OC2=CC=CC=C12)CN(C)C(=O)/C=C/C3=CC4=C(N=C3)N(C(=O)CC4)COP(=O)(O)O
|
|||||
| InChI |
1S/C23H24N3O7P/c1-15-18-5-3-4-6-19(18)33-20(15)13-25(2)21(27)9-7-16-11-17-8-10-22(28)26(23(17)24-12-16)14-32-34(29,30)31/h3-7,9,11-12H,8,10,13-14H2,1-2H3,(H2,29,30,31)/b9-7+
|
|||||
| InChIKey |
HFYMDQMXVPJNTH-VQHVLOKHSA-N
|
|||||
| CAS Number |
CAS 1518800-35-5
|
|||||
| PubChem Compound ID | ||||||
| Parent Drug General Information | Top | |||||
|---|---|---|---|---|---|---|
| Parent Drug ID |
D7Q5MA
|
|||||
| Parent Drug Name |
Debio 1452
|
|||||
| Synonyms |
AFN-1252; 620175-39-5; API-1252; UNII-T3O718IKKM; API 1252; T3O718IKKM; CHEMBL1652621; AFN-12520000; Debio 1452; afabicin desphosphono; DEBIO1452; SCHEMBL724936; SCHEMBL724937; DEBIO-1452; GTPL10755; DTXSID10211069; AFN-1252(Debio 1452); AFK-1252; BCP19135; BDBM50052244; ZINC38795123; DB12658; SB16787; NCGC00485479-01; AS-56008; HY-16911; Q27289625
Click to Show/Hide
|
|||||
| Formula |
C22H21N3O3
|
|||||
| Canonical SMILES |
CC1=C(OC2=CC=CC=C12)CN(C)C(=O)/C=C/C3=CC4=C(NC(=O)CC4)N=C3
|
|||||
| InChI |
1S/C22H21N3O3/c1-14-17-5-3-4-6-18(17)28-19(14)13-25(2)21(27)10-7-15-11-16-8-9-20(26)24-22(16)23-12-15/h3-7,10-12H,8-9,13H2,1-2H3,(H,23,24,26)/b10-7+
|
|||||
| InChIKey |
QXTWSUQCXCWEHF-JXMROGBWSA-N
|
|||||
| CAS Number |
CAS 620175-39-5
|
|||||
| PubChem Compound ID | ||||||
| Target and Pathway | Top | |||||
|---|---|---|---|---|---|---|
| Target(s) | Staphylococcus Enoyl ACP reductase (Stap-coc fabI) | Target Info | Inhibitor | [2] | ||
| KEGG Pathway | Fatty acid biosynthesis | |||||
| Biotin metabolism | ||||||
| Metabolic pathways | ||||||
| Fatty acid metabolism | ||||||
| References | Top | |||||
|---|---|---|---|---|---|---|
| REF 1 | ClinicalTrials.gov (NCT02426918) Study of Debio 1450 for Bacterial Skin Infections. U.S. National Institutes of Health. | |||||
| REF 2 | Prodrugs - Recent approvals and a glimpse of the pipeline. Eur J Pharm Sci. 2017 Nov 15;109:146-161. | |||||
| REF 3 | Preclinical pharmacokinetics and efficacy of debio 1450 (previously afn-1720), a prodrug of the staphylococcocal-specific antibiotic debio 1452 (previously afn-1252). | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

