Prodrug Information
| Prodrug General Information | Top | |||||
|---|---|---|---|---|---|---|
| Prodrug ID |
DKAQ01
|
|||||
| Prodrug Name |
Ceftobiprole medocaril
|
|||||
| Synonyms |
BAL5788; 376653-43-9; ceftobiprolemedocaril; Ceftobiprole Medocaril [INN]; SCHEMBL2671859; SCHEMBL19236293; DB14733; 653C439
Click to Show/Hide
|
|||||
| Indication | Methicillin-resistant staphylococci infection [ICD-11: 1A00-1A09; ICD-10: B95.62] | Approved in EU | [1] | |||
| Activation |
Prodrug 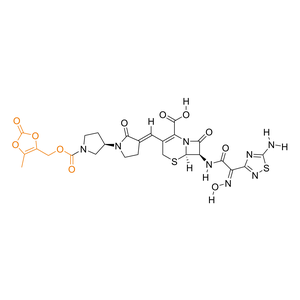
|

|
Parent Drug 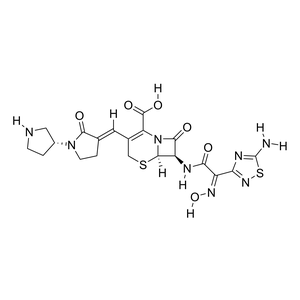
|
|||
| 2D MOL 3D MOL | 2D MOL 3D MOL | |||||
|
(1) Bioconversion Enzyme:
Esterase in plasma
(EC 3.1)
|
[2] | |||||
| Prodrug Strategy |
Classical prodrug strategy
[Carrier linked prodrug]
|
|||||
| Improved property |
Increase solubility; Prolong duration of action
|
[3] | ||||
| Formula |
C26H26N8O11S2
|
|||||
| Canonical SMILES |
CC1=C(OC(=O)O1)COC(=O)N2CC[C@H](C2)N3CC/C(=C\\C4=C(N5[C@@H]([C@@H](C5=O)NC(=O)/C(=N\\O)/C6=NSC(=N6)N)SC4)C(=O)O)/C3=O
|
|||||
| InChI |
1S/C26H26N8O11S2/c1-10-14(45-26(41)44-10)8-43-25(40)32-4-3-13(7-32)33-5-2-11(20(33)36)6-12-9-46-22-16(21(37)34(22)17(12)23(38)39)28-19(35)15(30-42)18-29-24(27)47-31-18/h6,13,16,22,42H,2-5,7-9H2,1H3,(H,28,35)(H,38,39)(H2,27,29,31)/b11-6+,30-15-/t13-,16-,22-/m1/s1
|
|||||
| InChIKey |
HFTSMHTWUFCYMJ-YIOMYIDASA-N
|
|||||
| CAS Number |
CAS 376653-43-9
|
|||||
| PubChem Compound ID | ||||||
| ChEBI ID |
CHEBI:135968
|
|||||
| Parent Drug General Information | Top | |||||
|---|---|---|---|---|---|---|
| Parent Drug ID |
DZYO54
|
|||||
| Parent Drug Name |
Ceftobiprole
|
|||||
| Synonyms |
BAL9141-000; Ro-63-9141; CHEMBL520642; ceftobiprol; ceftobiprolum; BAL-9141; Ro-639141; Ceftobiprole (USAN/INN); Ceftobiprole [USAN:INN]; SCHEMBL158871; SCHEMBL2671901; SCHEMBL19236292; DTXSID40870229; CHEBI:140407; DB04918; BAL-9141000; Ro 63-9141; BAL 9141-000; BAL-9141-000; HY-112579; CS-0047547; Ro-63-9141/000; D08885
Click to Show/Hide
|
|||||
| Formula |
C20H22N8O6S2
|
|||||
| Canonical SMILES |
C1CNC[C@@H]1N2CC/C(=C\\C3=C(N4[C@@H]([C@@H](C4=O)NC(=O)/C(=N\\O)/C5=NSC(=N5)N)SC3)C(=O)O)/C2=O
|
|||||
| InChI |
1S/C20H22N8O6S2/c21-20-24-14(26-36-20)11(25-34)15(29)23-12-17(31)28-13(19(32)33)9(7-35-18(12)28)5-8-2-4-27(16(8)30)10-1-3-22-6-10/h5,10,12,18,22,34H,1-4,6-7H2,(H,23,29)(H,32,33)(H2,21,24,26)/b8-5+,25-11-/t10-,12-,18-/m1/s1
|
|||||
| InChIKey |
VOAZJEPQLGBXGO-SDAWRPRTSA-N
|
|||||
| CAS Number |
CAS 209467-52-7
|
|||||
| PubChem Compound ID | ||||||
| ChEBI ID |
CHEBI:140407
|
|||||
| Target and Pathway | Top | |||||
|---|---|---|---|---|---|---|
| Target(s) | Bacterial Penicillin binding protein (Bact PBP) | Target Info | Binder | [1] | ||
| References | Top | |||||
|---|---|---|---|---|---|---|
| REF 1 | Ceftobiprole medocaril in the treatment of hospital-acquired pneumonia. Future Microbiol. 2015;10(12):1913-28. | |||||
| REF 2 | Ceftobiprole: A novel, broad-spectrum cephalosporin with activity against methicillin-resistant Staphylococcus aureus. Am J Health Syst Pharm. 2010 Jun 15;67(12):983-93. | |||||
| REF 3 | Ceftobiprole medocaril (BAL-5788) for the treatment of complicated skin infections. Expert Rev Anti Infect Ther. 2016;14(11):997-1006. | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

