Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D04SLF
|
||||
| Former ID |
DCL000497
|
||||
| Drug Name |
Bevacizumab + Trastuzumab
|
||||
| Drug Type |
Antibody
|
||||
| Company |
Roche
|
||||
| Structure |
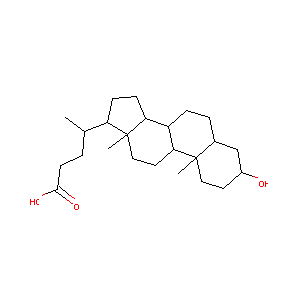
|
Download2D MOL |
|||
| Formula |
C24H40O3
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Vascular endothelial growth factor | Target Info | Inhibitor | [551607] | |
| Receptor protein-tyrosine kinase erbB-2 | Target Info | [551607] | |||
| KEGG Pathway | ErbB signaling pathway | ||||
| Calcium signaling pathway | |||||
| HIF-1 signaling pathway | |||||
| Focal adhesion | |||||
| Adherens junction | |||||
| Pathways in cancer | |||||
| Proteoglycans in cancer | |||||
| MicroRNAs in cancer | |||||
| Pancreatic cancer | |||||
| Endometrial cancer | |||||
| Prostate cancer | |||||
| Bladder cancer | |||||
| Non-small cell lung cancer | |||||
| Central carbon metabolism in cancer | |||||
| NetPath Pathway | TCR Signaling Pathway | ||||
| PANTHER Pathway | Cadherin signaling pathway | ||||
| EGF receptor signaling pathway | |||||
| PathWhiz Pathway | Phosphatidylinositol Phosphate Metabolism | ||||
| WikiPathways | DNA Damage Response (only ATM dependent) | ||||
| ErbB Signaling Pathway | |||||
| EGF/EGFR Signaling Pathway | |||||
| Focal Adhesion | |||||
| Extracellular vesicle-mediated signaling in recipient cells | |||||
| Bladder Cancer | |||||
| Signaling by ERBB2 | |||||
| Integrated Pancreatic Cancer Pathway | |||||
| Signaling Pathways in Glioblastoma | |||||
| Leptin signaling pathway | |||||
| miR-targeted genes in muscle cell - TarBase | |||||
| Semaphorin interactions | |||||
| References | |||||
| Ref 532301 | AVEREL: a randomized phase III Trial evaluating bevacizumab in combination with docetaxel and trastuzumab as first-line therapy for HER2-positive locally recurrent/metastatic breast cancer. J Clin Oncol. 2013 May 10;31(14):1719-25. | ||||
| Ref 554002 | ClinicalTrials.gov (NCT00365365) Safety & Efficacy of Three Docetaxel-Based Chemotherapy Regimens Plus Bevacizumab With or Without Trastuzumab for Adjuvant Treatment of Patients With Breast Cancer | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

