Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D07HVY
|
||||
| Former ID |
DAP000709
|
||||
| Drug Name |
Efavirenz
|
||||
| Synonyms |
EFV; EFZ; Eravirenz; Stocrin; Sustiva; DMP 266; L 743726; Atripla (TN); DMP-266; L-741211; L-743725; L-743726; Stocrin (TN); Strocin (TM); Sustiva (TM); Sustiva (TN); Efavirenz (JAN/INN); L-743,726; Zoxazin-2-one; Efavirenz, (S)-isomer; Met-SDF-1.beta. & Efavirenz; Met-Stromal Cell-derived Factor-1.beta. (Human) & Efavirenz; (-)-Efavirenz; (4S)-6-Chloro-4-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one; (4S)-6-Chloro-4-cyclopropylethynyl-4-trifluoromethyl-1,4-dihydro-benzo[d][1,3]oxazin-2-one; (4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-1H-3,1-benzoxazin-2-one; (4S)-6-chloro-4-(cyclopropylethynyl)-4-(trifluoromethyl)-1,4-dihydro-2H-3,1-benzoxazin-2-one; (S)-6-Chloro-4-(2-cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-ben; (S)-6-Chloro-4-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one; (S)-6-Chloro-4-cyclopropylethynyl-4-trifluoromethyl-1,4-dihydro-benzo[d][1,3]oxazin-2-one; 2H-3,1-Benzoxazin-2-one, 6-chloro-4-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-, (4S)-(9; 6-chloro-4-(2-cyclopropyl-1-ethynyl)-4-trifluoromethyl-(4S)-1,4-dihydro-2H-benzo[d][1,3]oxazin-2-one; EFV
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Human immunodeficiency virus infection [ICD9: 279.3; ICD10:B20-B26] | Approved | [550326] | ||
| Therapeutic Class |
Anti-HIV Agents
|
||||
| Structure |
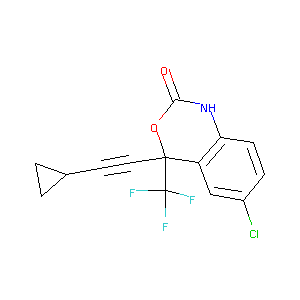
|
Download2D MOL |
|||
| Formula |
C14H9ClF3NO2
|
||||
| Canonical SMILES |
C1CC1C#CC2(C3=C(C=CC(=C3)Cl)NC(=O)O2)C(F)(F)F
|
||||
| InChI |
1S/C14H9ClF3NO2/c15-9-3-4-11-10(7-9)13(14(16,17)18,21-12(20)19-11)6-5-8-1-2-8/h3-4,7-8H,1-2H2,(H,19,20)/t13-/m0/s1
|
||||
| InChIKey |
XPOQHMRABVBWPR-ZDUSSCGKSA-N
|
||||
| CAS Number |
CAS 154598-52-4
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
10288, 615076, 644086, 819805, 819816, 822777, 822778, 823557, 2102933, 7847960, 7887287, 7979148, 8032270, 8189153, 12014858, 14850291, 26719858, 43121048, 46386724, 46392227, 46392228, 46506827, 49681705, 50064506, 53788925, 57315036, 78192089, 87350529, 91146581, 92308418, 92309272, 92717165, 93166541, 99444180, 104234240, 104330198, 118313744, 124658998, 126592964, 126608811, 126622440, 126654055, 129722732, 131300250, 134337998, 135022248, 136367956, 136903808, 137005170, 137229934
|
||||
| SuperDrug ATC ID |
J05AG03
|
||||
| SuperDrug CAS ID |
cas=154598524
|
||||
| Drug Resistance Mutation (DRM) | |||||
| DRM | DRM Info | ||||
| Target and Pathway | |||||
| Target(s) | HIV reverse transcriptase | Target Info | Modulator | [556264] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

