Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0KL3K
|
||||
| Former ID |
DIB000724
|
||||
| Drug Name |
YM-440
|
||||
| Synonyms |
2,2'-[2(Z)-Butene-1,4-diyl]dioxybis(1,4-phenylene)bis(methylene)bis[1,2,4-oxadiazole-3,5(2H,4H)-dione]
|
||||
| Indication | Type 1 diabetes [ICD9: 250; ICD10:E10] | Discontinued in Phase 2 | [546747] | ||
| Structure |
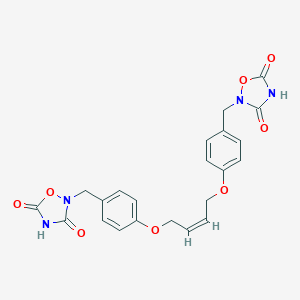
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Peroxisome proliferator activated receptor gamma | Target Info | Inhibitor | [528358], [551871] | |
| PANTHER Pathway | CCKR signaling map ST | ||||
| WikiPathways | Wnt Signaling Pathway Netpath | ||||
| Nuclear Receptors in Lipid Metabolism and Toxicity | |||||
| Differentiation of white and brown adipocyte | |||||
| Regulation of Lipid Metabolism by Peroxisome proliferator-activated receptor alpha (PPARalpha) | |||||
| Transcriptional Regulation of White Adipocyte Differentiation | |||||
| Adipogenesis | |||||
| SREBP signalling | |||||
| Nuclear Receptors | |||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

