COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D07EIQ
|
|||
| Drug Name |
Fluvoxamine
|
|||
| Synonyms |
fluvoxamine; 54739-18-3; Fluvoxaminum; Fluvoxamina; Fluvoxaminum [INN-Latin]; Fluvoxamina [INN-Spanish]; (Z)-Fluvoxamine; Fluvoxamine [INN:BAN]; UNII-O4L1XPO44W; C15H21F3N2O2; CHEMBL814; O4L1XPO44W; CHEBI:5138; DSSTox_RID_80097; DSSTox_CID_24002; DSSTox_GSID_44002; Fluvoxamine (INN); CAS; Fluvoxamine
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Coronavirus Disease 2019 (COVID-19) | Phase 2 | [1] | |
| Other Indication | Obsessive compulsive disorder | Approved | [2] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Structure |
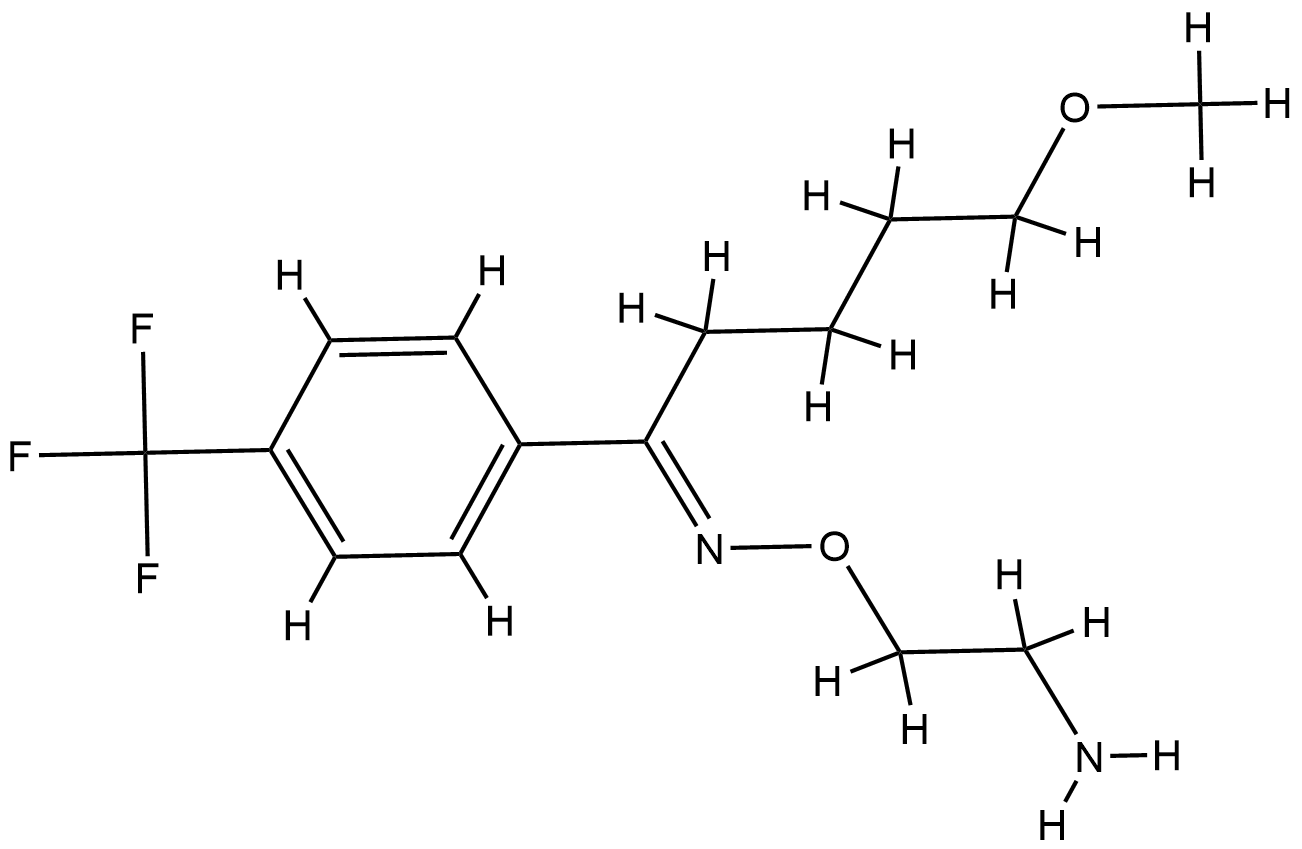 |
Download2D MOL |
||
| Formula |
C15H21F3N2O2
|
|||
| Canonical SMILES |
COCCCCC(=NOCCN)C1=CC=C(C=C1)C(F)(F)F
|
|||
| InChI |
1S/C15H21F3N2O2/c1-21-10-3-2-4-14(20-22-11-9-19)12-5-7-13(8-6-12)15(16,17)18/h5-8H,2-4,9-11,19H2,1H3/b20-14+
|
|||
| InChIKey |
CJOFXWAVKWHTFT-XSFVSMFZSA-N
|
|||
| CAS Number |
CAS 54739-18-3
|
|||
| PubChem Compound ID | ||||
| ChEBI ID |
CHEBI:5138
|
|||
| References | Top | |||
|---|---|---|---|---|
| 1 | ClinicalTrials.gov (NCT04342663) A Double-blind, Placebo-controlled Clinical Trial of Fluvoxamine for Symptomatic Individuals With COVID-19 Infection. U.S. National Institutes of Health. | |||
| 2 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (ANDA) 075893. | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

