COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D08ICX
|
|||
| Drug Name |
Tofacitinib
|
|||
| Synonyms |
Tofacitinib; Tasocitinib; CP-690550; CP 690550; racemic-tofacitinib; 1259404-17-5; CP-690,550; Tofacitinib (CP-690550,Tasocitinib); UNII-87LA6FU830; CHEMBL221959; CHEBI:71200; 87LA6FU830; 1-Piperi; Tofacitinib
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Coronavirus Disease 2019 (COVID-19) | Phase 2 | [1] | |
| Other Indication | Rheumatoid arthritis | Approved | [2] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Structure |
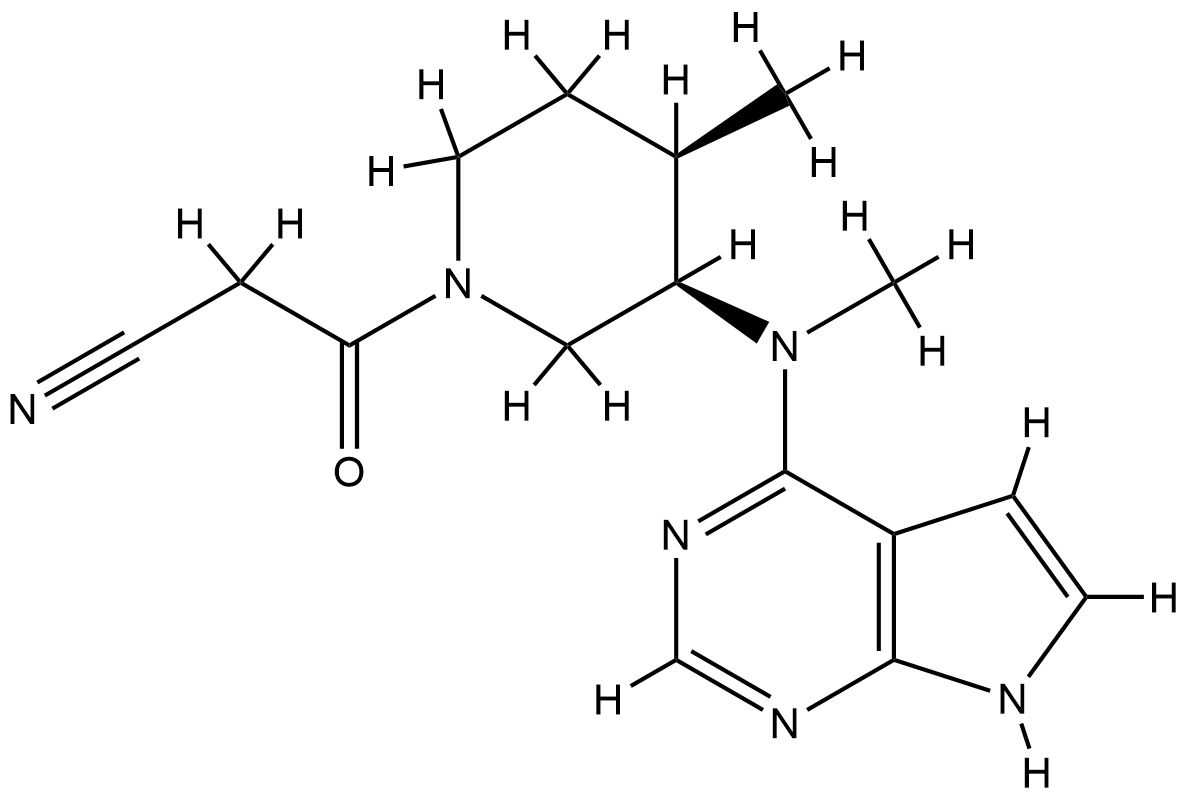 |
Download2D MOL |
||
| Formula |
C16H20N6O
|
|||
| Canonical SMILES |
CC1CCN(CC1N(C)C2=NC=NC3=C2C=CN3)C(=O)CC#N
|
|||
| InChI |
1S/C16H20N6O/c1-11-5-8-22(14(23)3-6-17)9-13(11)21(2)16-12-4-7-18-15(12)19-10-20-16/h4,7,10-11,13H,3,5,8-9H2,1-2H3,(H,18,19,20)/t11-,13+/m1/s1
|
|||
| InChIKey |
UJLAWZDWDVHWOW-YPMHNXCESA-N
|
|||
| CAS Number |
CAS 477600-75-2
|
|||
| PubChem Compound ID | ||||
| ChEBI ID |
CHEBI:71200
|
|||
| References | Top | |||
|---|---|---|---|---|
| 1 | ClinicalTrials.gov (NCT04332042) TOFAcitinib in SARS-CoV2 Pneumonia. U.S. National Institutes of Health. | |||
| 2 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 203214 | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

