COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D0K8RK
|
|||
| Drug Name |
Alisporivir
|
|||
| Synonyms |
DEB-025; Debio-025; HCV infection therapy, Debiopharm/Novartis; HIV infection therapy, Debiopharm
|
|||
| Drug Type |
Protein/peptide drug
|
|||
| Indication | Middle East Respiratory Syndrome (MERS) | Investigative | [1], [2] | Severe acute respiratory syndrome (SARS) | Investigative | [2] |
| Other Indication | Hepatitis C virus infection | Phase 3 | [3] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Company |
Novartis pharmaceuticals
|
|||
| Structure |
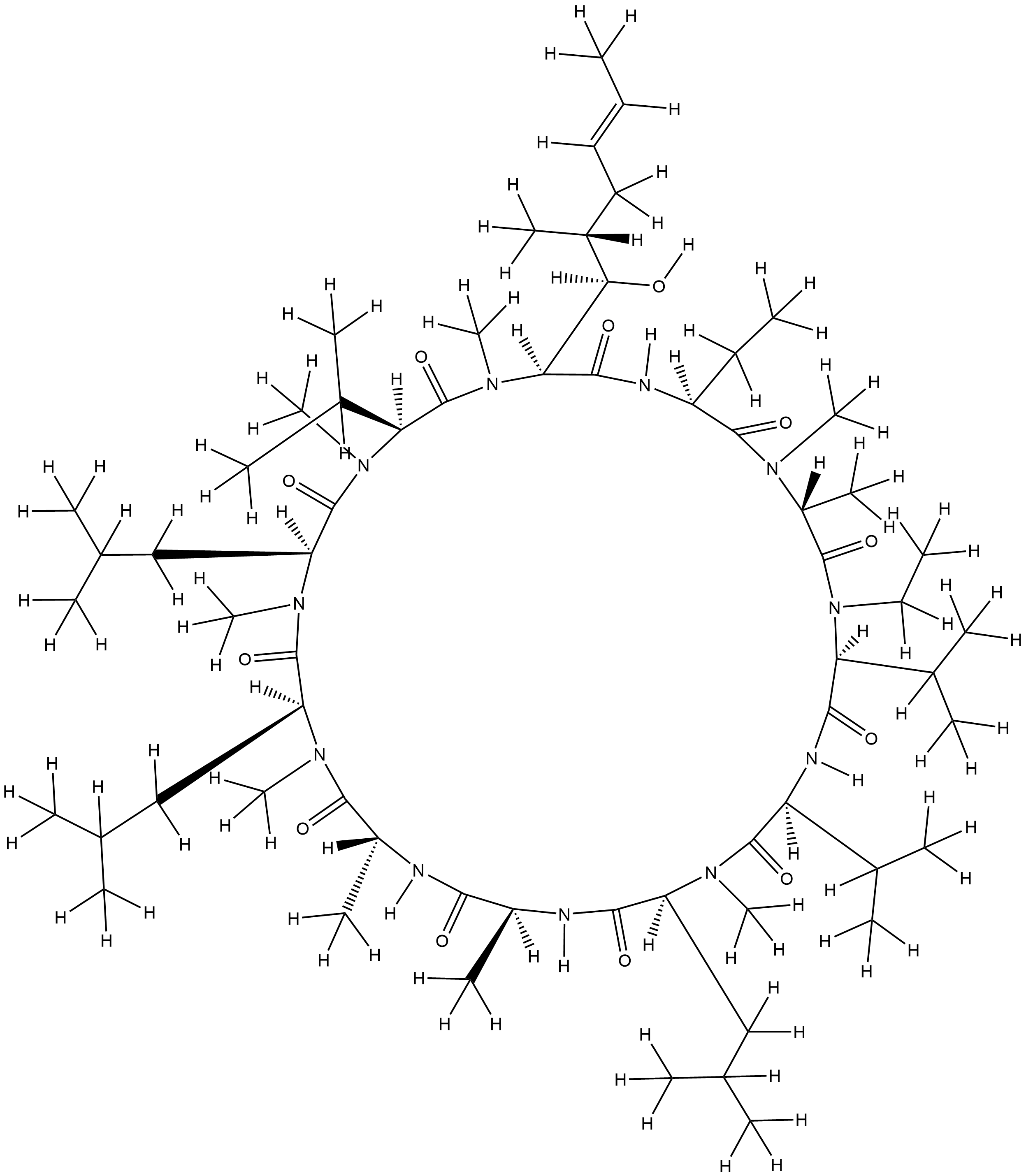 |
Download2D MOL
|
||
| Canonical SMILES |
CCC1C(=O)N(C(C(=O)N(C(C(=O)NC(C(=O)N(C(C(=O)NC(C(=O)NC(C(=O)N(C(C(=O)N(C(C(=O)N(C(C(=O)N(C(C(=O)N1)C(C(C)CC=CC)O)C)C(C)C)C)CC(C)C)C)CC(C)C)C)C)C)CC(C)C)C)C(C)C)C(C)C)CC)C)C
|
|||
| CAS Number |
CAS 254435-95-5
|
|||
| PubChem Compound ID | ||||
| Target | Top | |||
|---|---|---|---|---|
| Target(s) | HUMAN cyclophilin A (CYPA) | Target Info | Inhibitor | [1], [2] |
| Alisporivir inhibits a broad range of SARS-CoVs in vitro via inhibiting cyclophilin A that interact with SARS-CoV nsp1 to modulate the calcineurin pathway, which is important in the T cell-mediated adaptive immune response. | ||||
| References | Top | |||
|---|---|---|---|---|
| 1 | Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nat Rev Drug Discov. 2020 Mar;19(3):149-150. | |||
| 2 | Coronaviruses - drug discovery and therapeutic options. Nat Rev Drug Discov. 2016 May;15(5):327-47. | |||
| 3 | ClinicalTrials.gov (NCT01500772) Alisporivir With PEG and RBV in Protease Inhibitor (PI) Treatment Failure Patients With Chronic Hepatitis C | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

