COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D0M3TZ
|
|||
| Drug Name |
Mefloquine
|
|||
| Synonyms |
Lariam; Mefloquin; Mefloquina; Mefloquinone; Mefloquinum; Mephloquine; Racemic mefloquine; Ro 215998; WR 142490; Lariam (Hydrochloride); Lariam (TN); Mefaquin (TN); Mefloquina [INN-Spanish]; Mefloquinum [INN-Latin]; RO 13-7224; RO 13-7225; Ro 21-5998; SPB-80406; WR-142490; Mefloquine (USAN/INN); Mefloquine [USAN:INN:BAN]; RTI1169-1-1; RTI1172-1-1; RTI1173-1-1; RTI1174-1-1; RTI1188-1-1; RTI1189-1-1; Ro 21-5998 (Hydrochloride); WR-142,490; WR-177,602; Ro-21-5998-001; Alpha-2-Piperidinyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol; Alpha-2-Piperidyl-2,8-bis(trifluoromethyl)quinoline-4-methanol; [2,8-bis(trifluoromethyl)quinolin-4-yl]-piperidin-2-ylmethanol; Erthro-.alpha.-[2-piperidyl]-2,8-bis[trifluoromethyl]-4-quinolinemethanol; (+)-(11R,2'S)-erythro-Mefloquine; (+)-Mefloquine; (+)-Threo-Mefloquine; (-)-(11S,2'R)-erythro-Mefloquine; (-)-Mefloquine; (-)-Threo-Mefloquine; (DL-erythro-alpha-2-Piperidyl-2,8-bis(trifluoromethyl)-4-quinolinemethanol; (R)-[2,8-bis(trifluoromethyl)quinolin-4-yl]-[(2R)-piperidin-2-yl]methanol; (R)-[2,8-bis(trifluoromethyl)quinolin-4-yl]-[(2S)-piperidin-2-yl]methanol; (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl]-[(2R)-piperidin-2-yl]methanol; (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl]-[(2S)-piperidin-2-yl]methanol; (S)-[2,8-bis(trifluoromethyl)quinolin-4-yl][(2R)-piperidin-2-yl]methanol
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Coronavirus Disease 2019 (COVID-19) | Phase 2 | [1] | Middle East Respiratory Syndrome (MERS) | Preclinical | [2] | Severe acute respiratory syndrome (SARS) | Preclinical | [2] |
| Other Indication | Malaria | Approved | [3] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Company |
Hoffmann-La Roche pharmaceutical company
|
|||
| Structure |
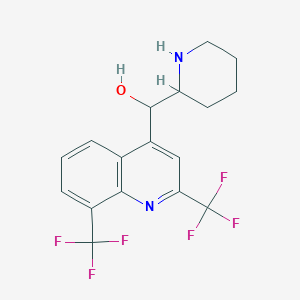 |
Download2D MOL |
||
| Formula |
C17H16F6N2O
|
|||
| Canonical SMILES |
C1CCNC(C1)C(C2=CC(=NC3=C2C=CC=C3C(F)(F)F)C(F)(F)F)O
|
|||
| InChI |
1S/C17H16F6N2O/c18-16(19,20)11-5-3-4-9-10(15(26)12-6-1-2-7-24-12)8-13(17(21,22)23)25-14(9)11/h3-5,8,12,15,24,26H,1-2,6-7H2
|
|||
| InChIKey |
XEEQGYMUWCZPDN-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 49752-90-1
|
|||
| PubChem Compound ID | ||||
| PubChem Substance ID |
9835, 603007, 8176741, 14877790, 26755467, 29216385, 34706437, 47206659, 49681220, 50042965, 50071333, 51051233, 53788688, 56310966, 56313823, 57312460, 85154870, 93166754, 93167184, 103446060, 104040016, 104335053, 124892361, 135004437, 135240375, 135611150, 135727482, 137236385, 140239432, 175269604, 178101069, 179116766, 223533437, 223712526, 226410988, 243866056, 252401319, 252655885
|
|||
| ChEBI ID |
CHEBI:63681
|
|||
| References | Top | |||
|---|---|---|---|---|
| 1 | ClinicalTrials.gov (NCT04347031) An Open Randomized Study of the Effectiveness of Mefloquine for the Treatment of Patients With COVID19. U.S. National Institutes of Health. | |||
| 2 | Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014 Aug;58(8):4885-93. | |||
| 3 | FDA Approved Drug Products from FDA Official Website. 2019. Application Number: (NDA) 019578 | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

