COVID-19 Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D0SR3J
|
|||
| Drug Name |
Loperamide
|
|||
| Synonyms |
Ioperamide; Loperacap; Loperamida; Loperamidum; Kaopectate II; Loperamide Monohydrochloride; Pepto Diarrhea Control; Apo-Loperamide; Diamide (TN); Diarr-Eze; Dimor (TN); Imodium (TN); Imodium A-D Caplets; Loperamida [INN-Spanish]; Loperamide (INN); Loperamide [INN:BAN]; Loperamidum [INN-Latin]; Lopex (TN); Maalox Anti-Diarrheal; Nu-Loperamide; PMS-Loperamide; Pepto (TN); R-18553; Rho-Loperamide
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Middle East Respiratory Syndrome (MERS) | Preclinical | [1] | Severe acute respiratory syndrome (SARS) | Preclinical | [1] |
| Other Indication | Diarrhea | Approved | [2] | |
| Therapeutic Class |
Antiviral Agents
|
|||
| Company |
Janssen Pharmaceutica
|
|||
| Structure |
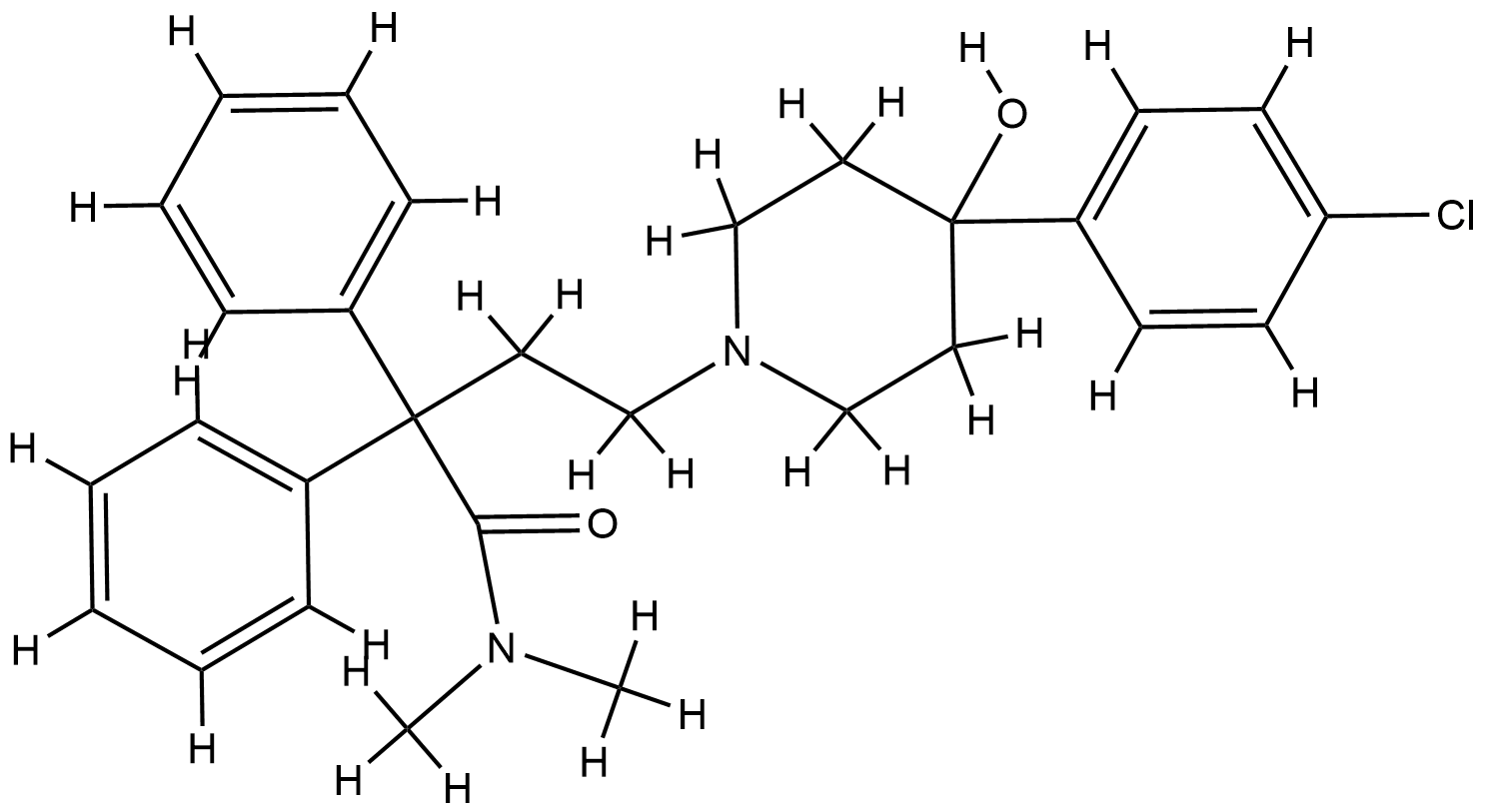 |
Download2D MOL |
||
| Formula |
C29H33ClN2O2
|
|||
| Canonical SMILES |
CN(C)C(=O)C(CCN1CCC(CC1)(C2=CC=C(C=C2)Cl)O)(C3=CC=CC=C3)C4=CC=CC=C4
|
|||
| InChI |
1S/C29H33ClN2O2/c1-31(2)27(33)29(24-9-5-3-6-10-24,25-11-7-4-8-12-25)19-22-32-20-17-28(34,18-21-32)23-13-15-26(30)16-14-23/h3-16,34H,17-22H2,1-2H3
|
|||
| InChIKey |
RDOIQAHITMMDAJ-UHFFFAOYSA-N
|
|||
| CAS Number |
CAS 53179-11-6
|
|||
| PubChem Compound ID | ||||
| PubChem Substance ID |
9291, 5686070, 7979790, 8152483, 10590039, 11111380, 11112740, 11113732, 11119963, 11120451, 11120939, 11121446, 11121926, 11147046, 11335390, 11360629, 11362515, 11363268, 11365077, 11365830, 11367639, 11368392, 11370301, 11370302, 11372088, 11373240, 11374823, 11375801, 11376554, 11461601, 11466172, 11467292, 11485552, 11485923, 11489554, 11490830, 11492957, 11494188, 14809921, 26751916, 26751917, 29223069, 46504591, 47216522, 47364910, 47515062, 47515063, 47588737, 47662002, 47662003
|
|||
| ChEBI ID |
CHEBI:6532
|
|||
| References | Top | |||
|---|---|---|---|---|
| 1 | Screening of an FDA-approved Compound Library Identifies Four Small-Molecule Inhibitors of Middle East Respiratory Syndrome Coronavirus Replication in Cell Culture Antimicrob Agents Chemother. 2014 Aug;58(8):4875-84. | |||
| 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

