Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D05DSK
|
||||
| Former ID |
DCL000282
|
||||
| Drug Name |
E2101
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Cervical dystonia; Spasm [ICD9: 333.83, 728.85; ICD10:G24.3, R25.2] | Discontinued in Phase 1 | [547174] | ||
| Company |
Eisai Co. Ltd.
|
||||
| Structure |
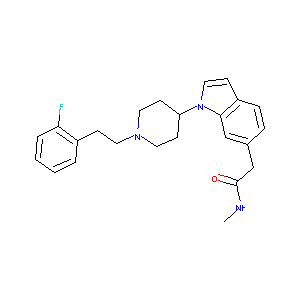
|
Download2D MOL |
|||
| Formula |
C24H28FN3O
|
||||
| Canonical SMILES |
CNC(=O)CC1=CC2=C(C=C1)C=CN2C3CCN(CC3)CCC4=CC=CC=C4F
|
||||
| InChI |
1S/C24H28FN3O/c1-26-24(29)17-18-6-7-20-9-15-28(23(20)16-18)21-10-13-27(14-11-21)12-8-19-4-2-3-5-22(19)25/h2-7,9,15-16,21H,8,10-14,17H2,1H3,(H,26,29)
|
||||
| InChIKey |
CQEOXWCXSBFJMB-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | 5-hydroxytryptamine 1A receptor | Target Info | Modulator | [535482] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

