Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D06BHG
|
||||
| Former ID |
DNCL002969
|
||||
| Drug Name |
Safotibant
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Diabetic macular edema [ICD9: 250, 362.01, 362.07, 362.53, 782.3; ICD10:E08-E13, E08.3, E09.3, E10.3, E11.3, E13.3, H35.8, R60.9] | Phase 2 | [523403] | ||
| Company |
Sanofi
|
||||
| Structure |
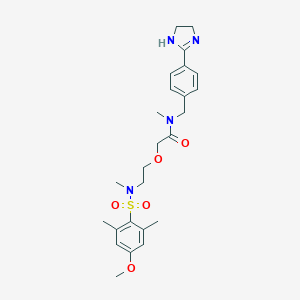
|
Download2D MOL |
|||
| Formula |
C25H34N4O5S
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | B1 bradykinin receptor | Target Info | Modulator | [551643] | |
| NetPath Pathway | Leptin Signaling Pathway | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

