Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0I1FQ
|
||||
| Former ID |
DAP001510
|
||||
| Drug Name |
Raltegravir
|
||||
| Synonyms |
RGV; MK 0518; Isentress(TM); K-0518; MK-0518; Raltegravir (INN); N-(2-(4-(4-fluorobenzylcarbamoyl); N-((4-Fluorophenyl)methyl)-1,6-dihydro-5-hydroxy-1-methyl-2-(1-methyl-1-(((5-methyl-1,3,4-oxadiazol-2-yl)carbonyl)amino)ethyl)-6-oxo-4-pyrimidinecarboxamide; RAL
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Human immunodeficiency virus infection [ICD9: 279.3; ICD10:B20-B26] | Approved | [529282] | ||
| Structure |
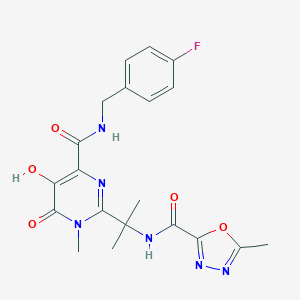
|
Download2D MOL |
|||
| Formula |
C20H21FN6O5
|
||||
| CAS Number |
CAS 871038-72-1
|
||||
| PubChem Compound ID | |||||
| SuperDrug ATC ID |
J05AX08
|
||||
| Drug Resistance Mutation (DRM) | |||||
| DRM | DRM Info | ||||
| Target and Pathway | |||||
| Target(s) | HIV integrase | Target Info | Modulator | [529282] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

