Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D00JED
|
||||
| Former ID |
DIB013295
|
||||
| Drug Name |
AN-2898
|
||||
| Synonyms |
PDE4 inhibitor (topical, psoriasis/atopic dermatitis), Anacor
|
||||
| Indication | Atopic dermatitis [ICD9: 691.8, 692.9; ICD10:L00-L99] | Phase 2 | [523369] | ||
| Company |
Anacor Pharmaceuticals Inc
|
||||
| Structure |
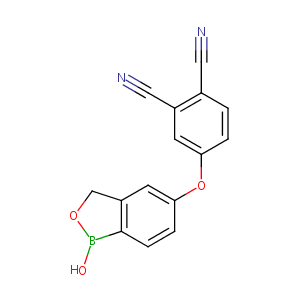
|
Download2D MOL |
|||
| Canonical SMILES |
c1(c(cc(cc1)Oc1cc2c(cc1)B(OC2)O)C#N)C#N
|
||||
| Target and Pathway | |||||
| Target(s) | Type IV phosphodiesterase | Target Info | Inhibitor | [532329] | |
| CAMP-specific 3',5'-cyclic phosphodiesterase 4B | Target Info | Inhibitor | [532329] | ||
| CAMP-specific 3',5'-cyclic phosphodiesterase 4A | Target Info | Inhibitor | [532329] | ||
| NetPath Pathway | IL5 Signaling Pathway | ||||
| IL2 Signaling Pathway | |||||
| PathWhiz Pathway | Purine Metabolism | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

