| Drug General Information |
| Drug ID |
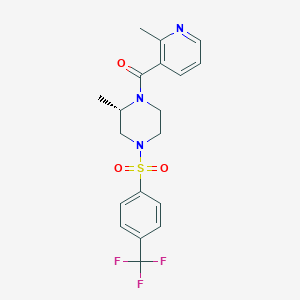
D01UDS
|
| Former ID |
DIB005017
|
| Drug Name |
CNV-2197944
|
| Synonyms |
GSK-2197944; Cav2.2 channel blocker (chronic pain), GlaxoSmithKline/Convergence Pharmaceuticals
|
| Indication |
Pain [ICD9: 338, 356.0, 356.8,780; ICD10:G64, G90.0, R52, G89]
|
Phase 2 |
[1]
|
|---|
| Company |
GlaxoSmithKline plc
|
| Structure |
|
Download
2D MOL
3D MOL
|
| Target and Pathway |
| Target(s) |
Voltage-dependent N-type calcium channel |
Target Info |
Blocker |
[2]
|
|---|
|
KEGG Pathway
|
MAPK signaling pathway
|
|
Calcium signaling pathway
|
|
Synaptic vesicle cycle
|
|
Retrograde endocannabinoid signaling
|
|
Cholinergic synapse
|
|
Serotonergic synapse
|
|
GABAergic synapse
|
|
Dopaminergic synapse
|
|
Taste transduction
|
|
Type II diabetes mellitus
|
|
Morphine addiction
|
|
Nicotine addiction
|
|
PANTHER Pathway
|
Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway
|
|
Ionotropic glutamate receptor pathway
|
|
Metabotropic glutamate receptor group III pathway
|
|
Metabotropic glutamate receptor group II pathway
|
|
Thyrotropin-releasing hormone receptor signaling pathway
|
|
Endogenous cannabinoid signaling
|
|
GABA-B receptor II signaling
|
|
Reactome
|
Depolarization of the Presynaptic Terminal Triggers the Opening of Calcium Channels
|
|
WikiPathways
|
Calcium Regulation in the Cardiac Cell
|
|
Synaptic Vesicle Pathway
|
| References |
| REF 1 | ClinicalTrials.gov (NCT01848730) Efficacy and Safety of CNV2197944 Versus Placebo in Patients With Post-herpetic Neuralgia. U.S. National Institutes of Health. |
|---|
| REF 2 | Clinical pipeline report, company report or official report of Convergence Pharmaceuticals Ltd. |
|---|