Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D08CZT
|
||||
| Former ID |
DNC003968
|
||||
| Drug Name |
2-Phenyl-3-piperidin-4-yl-1H-indole
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [1] | ||
| Structure |
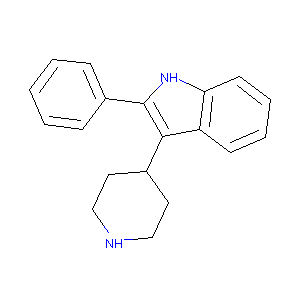
|
Download2D MOL |
|||
| Formula |
C19H20N2
|
||||
| Canonical SMILES |
C1CNCCC1C2=C(NC3=CC=CC=C32)C4=CC=CC=C4
|
||||
| InChI |
1S/C19H20N2/c1-2-6-15(7-3-1)19-18(14-10-12-20-13-11-14)16-8-4-5-9-17(16)21-19/h1-9,14,20-21H,10-13H2
|
||||
| InChIKey |
FWPAVENSJPMGJO-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Potassium voltage-gated channel subfamily H member 2 | Target Info | Inhibitor | [2] | |
| 5-hydroxytryptamine 2A receptor | Target Info | Inhibitor | [1] | ||
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Gap junction | |||||
| Serotonergic synapse | |||||
| Inflammatory mediator regulation of TRP channels | |||||
| PANTHER Pathway | 5HT2 type receptor mediated signaling pathway | ||||
| PathWhiz Pathway | Muscle/Heart Contraction | ||||
| Reactome | Voltage gated Potassium channelsR-HSA-390666:Serotonin receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | SIDS Susceptibility Pathways | ||||
| Hematopoietic Stem Cell Differentiation | |||||
| Potassium ChannelsWP733:Serotonin Receptor 2 and STAT3 Signaling | |||||
| Serotonin Receptor 2 and ELK-SRF/GATA4 signaling | |||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| GPCRs, Other | |||||
| References | |||||
| REF 1 | Bioorg Med Chem Lett. 2000 Dec 18;10(24):2701-3.3-(4-Piperidinyl)- and 3-(8-aza-bicyclo[3.2.1]oct-3-yl)-2-phenyl-1H-indoles as bioavailable h5-HT2A antagonists. | ||||
| REF 2 | J Med Chem. 2001 May 10;44(10):1603-14.3-(4-Fluoropiperidin-3-yl)-2-phenylindoles as high affinity, selective, and orally bioavailable h5-HT(2A) receptor antagonists. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

