Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D08UGX
|
||||
| Former ID |
DNC008925
|
||||
| Drug Name |
LIAROZOLE
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Dermatological disease [ICD10:L00-L99] | Phase 2/3 | [1], [2] | ||
| Structure |
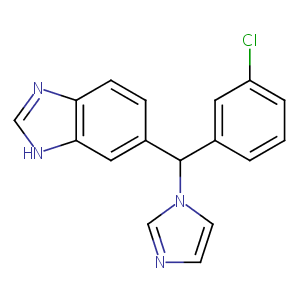
|
Download2D MOL |
|||
| Formula |
C17H13ClN4
|
||||
| InChI |
InChI=1S/C17H13ClN4/c18-14-3-1-2-12(8-14)17(22-7-6-19-11-22)13-4-5-15-16(9-13)21-10-20-15/h1-11,17H,(H,20,21)
|
||||
| InChIKey |
UGFHIPBXIWJXNA-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Cytochrome P450 26 | Target Info | Inhibitor | [3] | |
| Cytochrome P450 19 | Target Info | Inhibitor | [4] | ||
| BioCyc Pathway | Superpathway of steroid hormone biosynthesis | ||||
| Estradiol biosynthesis II | |||||
| Estradiol biosynthesis I | |||||
| KEGG Pathway | Retinol metabolism | ||||
| Metabolic pathwayshsa00140:Steroid hormone biosynthesis | |||||
| Metabolic pathways | |||||
| Ovarian steroidogenesis | |||||
| NetPath Pathway | FSH Signaling Pathway | ||||
| PANTHER Pathway | Androgen/estrogene/progesterone biosynthesis | ||||
| PathWhiz Pathway | Retinol MetabolismPW000045:Androgen and Estrogen Metabolism | ||||
| Reactome | Endogenous sterols | ||||
| WikiPathways | Vitamin A and Carotenoid Metabolism | ||||
| Metapathway biotransformation | |||||
| Oxidation by Cytochrome P450 | |||||
| Nuclear Receptors in Lipid Metabolism and Toxicity | |||||
| Adipogenesis | |||||
| Phase 1 - Functionalization of compoundsWP702:Metapathway biotransformation | |||||
| Tryptophan metabolism | |||||
| Ovarian Infertility Genes | |||||
| Metabolism of steroid hormones and vitamin D | |||||
| FSH signaling pathway | |||||
| Integrated Breast Cancer Pathway | |||||
| Phase 1 - Functionalization of compounds | |||||
| References | |||||
| REF 1 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 5210). | ||||
| REF 2 | ClinicalTrials.gov (NCT00282724) Efficacy and Safety of Two Doses of Liarozole vs. Placebo for the Treatment of Lamellar Ichthyosis. U.S. National Institutes of Health. | ||||
| REF 3 | Bioorg Med Chem. 2008 Sep 1;16(17):8301-13. Epub 2007 Jun 29.Novel azolyl-(phenylmethyl)]aryl/heteroarylamines: potent CYP26 inhibitors and enhancers of all-trans retinoic acid activity in neuroblastoma cells. | ||||
| REF 4 | Bioorg Med Chem Lett. 2010 May 15;20(10):3050-64. Epub 2010 Apr 8.Pharmacophore modeling strategies for the development of novel nonsteroidal inhibitors of human aromatase (CYP19). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

