Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0H0UB
|
||||
| Former ID |
DCL000515
|
||||
| Drug Name |
Darotropium + 642444
|
||||
| Indication | Chronic obstructive pulmonary disease [ICD9: 490-492, 494-496; ICD10:J40-J44, J47] | Phase 3 | [523399] | ||
| Company |
GSK
|
||||
| Structure |
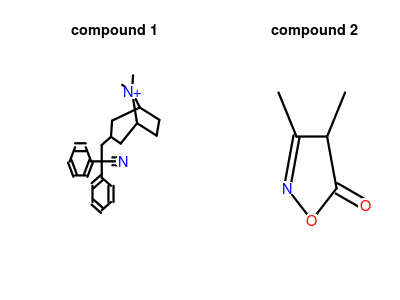
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Muscarinic acetylcholine receptor M1 | Target Info | Agonist | [550963] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

