Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0GE8S
|
||||
| Former ID |
DNC007864
|
||||
| Drug Name |
ES-936
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [529109] | ||
| Structure |
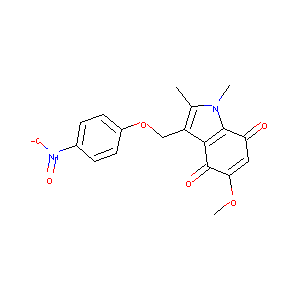
|
Download2D MOL |
|||
| Formula |
C18H16N2O6
|
||||
| Canonical SMILES |
CC1=C(C2=C(N1C)C(=O)C=C(C2=O)OC)COC3=CC=C(C=C3)[N+](=O)<br />[O-]
|
||||
| InChI |
1S/C18H16N2O6/c1-10-13(9-26-12-6-4-11(5-7-12)20(23)24)16-17(19(10)2)14(21)8-15(25-3)18(16)22/h4-8H,9H2,1-3H3
|
||||
| InChIKey |
IBLWSLZYYZHSRG-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | NAD(P)H dehydrogenase [quinone] 1 | Target Info | Inhibitor | [529109] | |
| KEGG Pathway | Ubiquinone and other terpenoid-quinone biosynthesis | ||||
| NetPath Pathway | TCR Signaling Pathway | ||||
| Pathway Interaction Database | Validated transcriptional targets of TAp63 isoforms | ||||
| PathWhiz Pathway | Vitamin K Metabolism | ||||
| WikiPathways | Estrogen metabolism | ||||
| Oxidative Stress | |||||
| Transcriptional activation by NRF2 | |||||
| NRF2 pathway | |||||
| Nuclear Receptors Meta-Pathway | |||||
| Aryl Hydrocarbon Receptor Pathway | |||||
| Apoptosis-related network due to altered Notch3 in ovarian cancer | |||||
| Metabolism of amino acids and derivatives | |||||
| Aryl Hydrocarbon Receptor | |||||
| Dopamine metabolism | |||||
| Arylhydrocarbon receptor (AhR) signaling pathway | |||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

