Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0L6VH
|
||||
| Former ID |
DIB005930
|
||||
| Drug Name |
BF-389
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Rheumatoid arthritis [ICD9: 710-719, 714; ICD10:M05-M06] | Phase 1 | [1] | ||
| Company |
Biofor Inc
|
||||
| Structure |
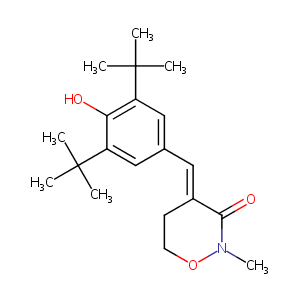
|
Download2D MOL |
|||
| Formula |
C20H29NO3
|
||||
| Canonical SMILES |
C1(=O)/C(=C/c2cc(c(c(C(C)(C)C)c2)O)C(C)(C)C)/CCON1C
|
||||
| CAS Number |
CAS 127245-22-1
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Arachidonate 5-lipoxygenase | Target Info | Modulator | [1] | |
| BioCyc Pathway | Aspirin-triggered lipoxin biosynthesis | ||||
| Resolvin D biosynthesis | |||||
| Leukotriene biosynthesis | |||||
| Lipoxin biosynthesis | |||||
| Aspirin triggered resolvin D biosynthesis | |||||
| Aspirin triggered resolvin E biosynthesis | |||||
| KEGG Pathway | Arachidonic acid metabolism | ||||
| Metabolic pathways | |||||
| Serotonergic synapse | |||||
| Ovarian steroidogenesis | |||||
| Toxoplasmosis | |||||
| NetPath Pathway | IL4 Signaling Pathway | ||||
| PathWhiz Pathway | Arachidonic Acid Metabolism | ||||
| WikiPathways | Vitamin D Receptor Pathway | ||||
| Arachidonic acid metabolism | |||||
| Eicosanoid Synthesis | |||||
| Selenium Micronutrient Network | |||||
| References | |||||
| REF 1 | Antiarthritic profile of BF-389--a novel anti-inflammatory agent with low ulcerogenic liability. Agents Actions. 1992 Sep;37(1-2):90-8. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

