Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0KV4Z
|
||||
| Former ID |
DIB008978
|
||||
| Drug Name |
CI-949
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Asthma [ICD10:J45] | Discontinued in Phase 2 | [1] | ||
| Company |
Dura Pharmaceuticals Inc
|
||||
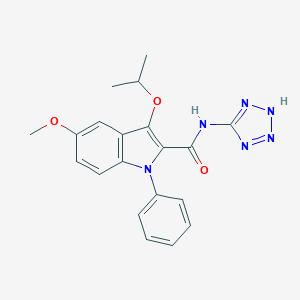
| Structure |
|
Download2D MOL |
|||
| Formula |
C26H34N10O5
|
||||
| Canonical SMILES |
c1(n(c2c(c1OC(C)C)cc(cc2)OC)c1ccccc1)C(=O)Nc1nnn[nH]1.C<br />(=N)(NCCC[C@@H](C(=O)O)N)N
|
||||
| CAS Number |
CAS 121530-58-3
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Leukotriene B4 receptor 1 | Target Info | Modulator | [2] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| NetPath Pathway | IL4 Signaling Pathway | ||||
| Reactome | Leukotriene receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Nucleotide GPCRs | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Spinal Cord Injury | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800000104) | ||||
| REF 2 | Inhibition of histamine, leukotriene C4/D4, and thromboxane B2 release from human leukocytes and human chopped lung mast cells by the allergic mediator release inhibitor, CI-949. J Allergy Clin Immunol. 1990 Dec;86(6 Pt 1):902-8. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

