Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D4LOR2
|
|||
| Drug Name |
Tebipenem pivoxil
|
|||
| Synonyms |
161715-24-8; Orapenem; TBPM-PI; L-084; ME1211; UNII-95AK1A52I8; LJC 11,084; 95AK1A52I8; LJC-11084; ME-1211; 2,2-dimethylpropanoyloxymethyl (4R,5S,6S)-3-[1-(4,5-dihydro-1,3-thiazol-2-yl)azetidin-3-yl]sulfanyl-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate; Tebipenempivoxil; Tebipenem [INN]; (4R,5S,6S)-(Pivaloyloxy)methyl 3-((1-(4,5-dihydrothiazol-2-yl)azetidin-3-yl)thio)-6-((R)-1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate; 1-Azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid, 3-[[1-(4,5-dihydro-2-thiazolyl)-3-azetidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-, (2,2-dimethyl-1-oxopropoxy)methyl ester, (4R,5S,6S)-; Tebipenem pivoxil (L-084); Tebipenem pivoxil(L-084); Orapenem (TN); (1R,5S,6S)-6-[1(R)-Hydroxyethyl]-1-methyl-2-[1-(2-thiazolin-2-yl)azetidin-3-ylsulfanyl]-1-carba-2-penem-3-carboxylic acid pivaloyloxymethyl ester; MLS006011128; SCHEMBL305541; Tebipenem pivoxil [USAN:INN]; CHEMBL2107486; GTPL10864; DTXSID00167228; CHEBI:135799; EX-A1256; HY-B0396; WHO 7924; ZINC4217736; Tebipenem pivoxil (JAN/USAN/INN); 2748AH; CT0255; LJC 11084; MFCD17215369; s2159; AKOS015899532; CCG-269673; LJC-11,084; NCGC00346570-01; NCGC00346570-04; (pivaloyloxy)methyl (4R,5S,6S)-3-((1-(4,5-dihydrothiazol-2-yl)azetidin-3-yl)thio)-6-((R)-1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate; [(4R,5S,6S)-3-{[1-(4,5-dihydro-1,3-thiazol-2-yl)azetidin-3-yl]sulfanyl}-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carbonyloxy]methyl 2,2-dimethylpropanoate; AC-22712; AS-75334; L-084;ME1211; SMR004702908; L 084; D09598; AB01566835_01; Q1623593; (4R,5R,6S)-3-((1-(4,5-Dihydro-2-thiazolyl)-3-azetidinyl)thio)-6-((1R)-1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo(3.2.0)hept-2-ene-2-carboxylic acid (2,2-dimethyl-1-oxopropoxy) methyl ester; 2,2-dimethylpropanoyloxymethyl (4R,5S,6S)-3-[1-(4,5-dihydrothiazol-2-yl)azetidin-3-yl]sulfanyl-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate
Click to Show/Hide
|
|||
| Drug Type |
Small molecular drug
|
|||
| Indication | Respiratory tract infection [ICD-11: CA45; ICD-10: J13, J15] | Approved | [1] | |
| Company |
Meiji Seika Pharma
|
|||
| Structure |
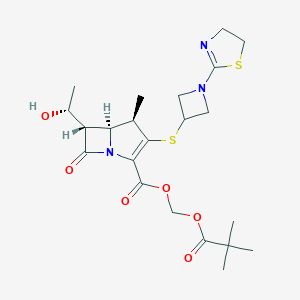 |
Download2D MOL |
||
| Formula |
C22H31N3O6S2
|
|||
| Canonical SMILES |
CC1C2C(C(=O)N2C(=C1SC3CN(C3)C4=NCCS4)C(=O)OCOC(=O)C(C)(C)C)C(C)O
|
|||
| InChI |
1S/C22H31N3O6S2/c1-11-15-14(12(2)26)18(27)25(15)16(19(28)30-10-31-20(29)22(3,4)5)17(11)33-13-8-24(9-13)21-23-6-7-32-21/h11-15,26H,6-10H2,1-5H3/t11-,12-,14-,15-/m1/s1
|
|||
| InChIKey |
SNUDIPVBUUXCDG-QHSBEEBCSA-N
|
|||
| CAS Number |
CAS 161715-24-8
|
|||
| PubChem Compound ID | ||||
| ChEBI ID |
CHEBI:135799
|
|||
| Target and Pathway | Top | |||
|---|---|---|---|---|
| Target(s) | Bacterial Penicillin binding protein (Bact PBP) | Target Info | Binder | [2] |
| References | Top | |||
|---|---|---|---|---|
| REF 1 | Pharmacodynamics of Tebipenem: New Options for Oral Treatment of Multidrug-Resistant Gram-Negative Infections. Antimicrob Agents Chemother. 2019 Jul 25;63(8):e00603-19. | |||
| REF 2 | In Vitro Activity of Tebipenem (SPR859) against Penicillin-Binding Proteins of Gram-Negative and Gram-Positive Bacteria. Antimicrob Agents Chemother. 2019 Mar 27;63(4):e02181-18. | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

