Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0B0QY
|
||||
| Former ID |
DNC008920
|
||||
| Drug Name |
5(E)-(5'-Deoxyadenosin-5'-ylidene)pentanoic acid
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [1] | ||
| Structure |
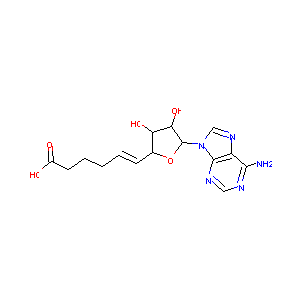
|
Download2D MOL |
|||
| Formula |
C15H19N5O5
|
||||
| Canonical SMILES |
C1=NC2=C(C(=N1)N)N=CN2C3C(C(C(O3)C=CCCCC(=O)O)O)O
|
||||
| InChI |
1S/C15H19N5O5/c16-13-10-14(18-6-17-13)20(7-19-10)15-12(24)11(23)8(25-15)4-2-1-3-5-9(21)22/h2,4,6-8,11-12,15,23-24H,1,3,5H2,(H,21,22)(H2,16,17,18)/b4-2+/t8-,11-,12-,15-/m1/s1
|
||||
| InChIKey |
HAGCQDHTBHGTQC-VLCWARKCSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Adenosylhomocysteinase | Target Info | Inhibitor | [1] | |
| BioCyc Pathway | Superpathway of methionine degradation | ||||
| Methionine degradation | |||||
| Cysteine biosynthesis | |||||
| KEGG Pathway | Cysteine and methionine metabolism | ||||
| Metabolic pathways | |||||
| PathWhiz Pathway | Selenoamino Acid Metabolism | ||||
| Betaine Metabolism | |||||
| Methionine Metabolism | |||||
| WikiPathways | Metabolism of amino acids and derivatives | ||||
| Trans-sulfuration and one carbon metabolism | |||||
| One Carbon Metabolism | |||||
| Trans-sulfuration pathway | |||||
| Phase II conjugation | |||||
| Folate Metabolism | |||||
| References | |||||
| REF 1 | Bioorg Med Chem. 2008 May 15;16(10):5424-33. Epub 2008 Apr 12.Synthesis of 5'-functionalized nucleosides: S-Adenosylhomocysteine analogues with the carbon-5' and sulfur atoms replaced by a vinyl or halovinyl unit. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

