Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0Z9GM
|
||||
| Former ID |
DIB004379
|
||||
| Drug Name |
LY-2940094
|
||||
| Indication | Major depressive disorder [ICD9: 296.2, 296.3, 710.0; ICD10:F32, F33, M32] | Phase 2 | [524117] | ||
| Company |
Eli Lilly & Co
|
||||
| Structure |
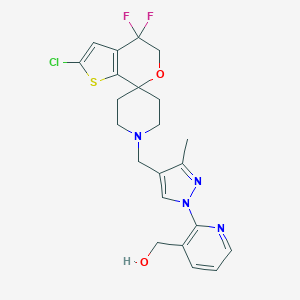
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Nociceptin receptor | Target Info | Modulator | [551571], [551633] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

