Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D01HZE
|
||||
| Former ID |
DNC012749
|
||||
| Drug Name |
BRL-25594
|
||||
| Indication | Discovery agent | Investigative | [1] | ||
| Structure |
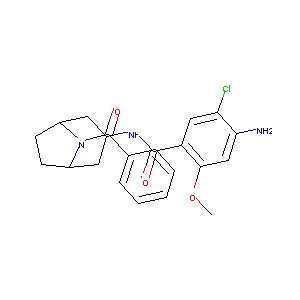
|
Download2D MOL |
|||
| Canonical SMILES |
COC1=CC(=C(C=C1C(=O)NC2CC3CCC(C2)N3C(=O)C4=CC=CC=C4)Cl)<br />N
|
||||
| InChI |
1S/C22H24ClN3O3/c1-29-20-12-19(24)18(23)11-17(20)21(27)25-14-9-15-7-8-16(10-14)26(15)22(28)13-5-3-2-4-6-13/h2-6,11-12,14-16H,7-10,24H2,1H3,(H,25,27)
|
||||
| InChIKey |
YFYKDXUABDOBBU-UHFFFAOYSA-N
|
||||
| Target and Pathway | |||||
| Target(s) | D(2) dopamine receptor | Target Info | Inhibitor | [1] | |
| KEGG Pathway | Rap1 signaling pathway | ||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| Gap junction | |||||
| Dopaminergic synapse | |||||
| Parkinson's disease | |||||
| Cocaine addiction | |||||
| Alcoholism | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | |||||
| Dopamine receptor mediated signaling pathway | |||||
| Nicotine pharmacodynamics pathway | |||||
| Reactome | Dopamine receptors | ||||
| G alpha (i) signalling events | |||||
| WikiPathways | Hypothetical Network for Drug Addiction | ||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Genes and (Common) Pathways Underlying Drug Addiction | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| Nicotine Activity on Dopaminergic Neurons | |||||
| References | |||||
| REF 1 | J Med Chem. 2003 Feb 27;46(5):702-15.Synthesis and structure-affinity relationships of novel N-(1-ethyl-4-methylhexahydro-1,4-diazepin-6-yl)pyridine-3-carboxamides with potent serotonin 5-HT3 and dopamine D2 receptor antagonistic activity. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

