Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0T1XW
|
||||
| Former ID |
DAP000795
|
||||
| Drug Name |
Meclizine
|
||||
| Synonyms |
Ancolan; Ancolon; Antivert; Bonadettes; Bonine; Calmonal; Chiclida; Diadril; Histamethine; Histamethizine; Histametizine; Histametizyn; Histametizyne; Itinerol; Marex; Meclicot; Meclozina; Meclozine; Meclozinum; Medivert; Monamine; Navicalm; Nevidoxine; Parachloramine; Peremesin; Postafene; Ravelon; Sabari; Siguran; Subari; Suprimal; Travelon; Veritab; Vomisseis; Vomissels; Dramamine II; Meclizine Hcl; UCB 170; UCB 5052; UCB 5062; Antivert (TN); Antivert/25; Antivert/50; Bonamine (TN); Bonine (TN); Dramamine (TN); Meclizine [INN:BAN]; Meclozina [INN-Spanish]; Meclozine (BAN); Meclozinum [INN-Latin]; Neo-istafene; Neo-suprimal; Neo-suprimel; Nevidoxine (TN); Postafen (TN); Sea-Legs; Ru-Vert-M; U. C. B. 5062; U.C.B. 5062; (+-)-Meclizine; 1-((4-Chlorophenyl)phenylmethyl)-4-((3-methylphenyl)methyl)piperazine; 1-(p-Chloro-.alpha.-phenylbenzyl)-4-(m-methylbenzyl)piperazine; 1-(p-Chloro-alpha-phenylbenzyl)-4-(m-methylbenzyl)piperazine; 1-(p-Chlorobenzhydryl)-4-(m-methylbenzyl)diethylenediamine; 1-(p-Chlorobenzhydryl)-4-(m-methylbenzyl)piperazine; 1-[(4-chlorophenyl)(phenyl)methyl]-4-[(3-methylphenyl)methyl]piperazine; 1-[(4-chlorophenyl)-phenylmethyl]-4-[(3-methylphenyl)methyl]piperazine; 1-p-Chlorobenzhydryl-4-m-methylbenzylpiperazine
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Nausea; Vomiting; Dizziness [ICD10:R11] | Approved | [551871] | ||
| Therapeutic Class |
Antiallergic Agents
|
||||
| Company |
Citron Pharma Llc
|
||||
| Structure |
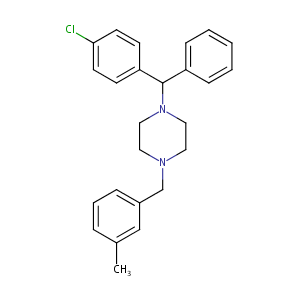
|
Download2D MOL |
|||
| Formula |
C25H27ClN2
|
||||
| InChI |
InChI=1S/C25H27ClN2/c1-20-6-5-7-21(18-20)19-27-14-16-28(17-15-27)25(22-8-3-2-4-9-22)23-10-12-24(26)13-11-23/h2-13,18,25H,14-17,19H2,1H3
|
||||
| InChIKey |
OCJYIGYOJCODJL-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 569-65-3
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
9327, 442158, 4782133, 7979876, 8152525, 10527723, 11335582, 11360821, 11363785, 11366347, 11368909, 11371549, 11373612, 11377071, 11461793, 11466485, 11467605, 11483836, 11486209, 11487926, 11490301, 11491852, 11494705, 14805236, 29223145, 46507782, 47440184, 47515252, 47810681, 47959664, 48035039, 48110390, 48334421, 48416201, 49698867, 50012193, 50807781, 57322105, 76715647, 85209822, 85788825, 96024853, 103604960, 104305237, 117409993, 124883625, 124883627, 125312386, 125823630, 126689207
|
||||
| SuperDrug ATC ID |
R06AE05
|
||||
| SuperDrug CAS ID |
cas=000569653
|
||||
| Target and Pathway | |||||
| Target(s) | Orphan nuclear receptor NR1I3 | Target Info | Modulator | [536227] | |
| Pathway Interaction Database | Glucocorticoid receptor regulatory network | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

