Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0UI3T
|
||||
| Former ID |
DNCL002721
|
||||
| Drug Name |
Elagolix
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Uterine leiomyoma [ICD9: 218.9; ICD10:D25] | Phase 3 | [1], [2] | ||
| Company |
Abbott Laboratories Abbott; Neurocrine Biosciences
|
||||
| Structure |
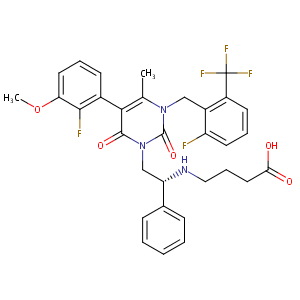
|
Download2D MOL |
|||
| Formula |
C32H30F5N3O5
|
||||
| InChI |
InChI=1S/C32H30F5N3O5/c1-19-28(21-11-6-14-26(45-2)29(21)34)30(43)40(18-25(20-9-4-3-5-10-20)38-16-8-15-27(41)42)31(44)39(19)17-22-23(32(35,36)37)12-7-13-24(22)33/h3-7,9-14,25,38H,8,15-18H2,1-2H3,(H,41,42)/t25-/m0/s1
|
||||
| InChIKey |
HEAUOKZIVMZVQL-VWLOTQADSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Gonadotropin-releasing hormone receptor | Target Info | Antagonist | [3] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| GnRH signaling pathway | |||||
| NetPath Pathway | IL1 Signaling Pathway | ||||
| IL2 Signaling Pathway | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | ||||
| Reactome | Hormone ligand-binding receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Gastrin-CREB signalling pathway via PKC and MAPK | ||||
| Peptide GPCRs | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| GPCRs, Other | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT01620528) A Clinical Study to Evaluate the Safety and Efficacy of Elagolix in Subjects With Moderate to Severe Endometriosis-Associated Pain. U.S. National Institutes of Health. | ||||
| REF 2 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 8362). | ||||
| REF 3 | Elagolix, a novel, orally bioavailable GnRH antagonist under investigation for the treatment of endometriosis-related pain. Womens Health (Lond Engl). 2015 Jan;11(1):19-28. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

