Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D06GQY
|
||||
| Former ID |
DNC010488
|
||||
| Drug Name |
PSB-0963
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [530711] | ||
| Structure |
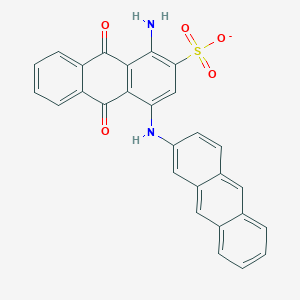
|
Download2D MOL |
|||
| Formula |
C28H17N2NaO5S
|
||||
| Canonical SMILES |
C1=CC=C2C=C3C=C(C=CC3=CC2=C1)NC4=CC(=C(C5=C4C(=O)C6=CC=<br />CC=C6C5=O)N)S(=O)(=O)[O-].[Na+]
|
||||
| InChI |
1S/C28H18N2O5S.Na/c29-26-23(36(33,34)35)14-22(24-25(26)28(32)21-8-4-3-7-20(21)27(24)31)30-19-10-9-17-11-15-5-1-2-6-16(15)12-18(17)13-19;/h1-14,30H,29H2,(H,33,34,35);/q;+1/p-1
|
||||
| InChIKey |
YFKPIPRCPCDQAK-UHFFFAOYSA-M
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | P2Y purinoceptor 2 | Target Info | Inhibitor | [530711] | |
| Ectonucleoside triphosphate diphosphohydrolase 1 | Target Info | Inhibitor | [530711] | ||
| 5'-Nucleotidase | Target Info | Inhibitor | [530711] | ||
| NetPath Pathway | IL5 Signaling Pathway | ||||
| PANTHER Pathway | Purine metabolism | ||||
| Pyrimidine Metabolism | |||||
| Pathway Interaction Database | HIF-1-alpha transcription factor network | ||||
| WikiPathways | Nucleotide GPCRs | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signalingWP2848:Differentiation Pathway | |||||
| miR-targeted genes in muscle cell - TarBase | |||||
| miR-targeted genes in lymphocytes - TarBase | |||||
| miR-targeted genes in epithelium - TarBase | |||||
| Metabolism of nucleotides | |||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

