Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0A6AX
|
||||
| Former ID |
DNC011402
|
||||
| Drug Name |
(9H-beta-Carbolin-3-yl)-carbamic acid ethyl ester
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [1] | ||
| Structure |
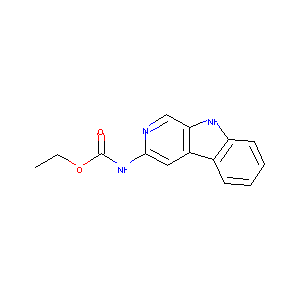
|
Download2D MOL |
|||
| Formula |
C14H13N3O2
|
||||
| Canonical SMILES |
CCOC(=O)NC1=NC=C2C(=C1)C3=CC=CC=C3N2
|
||||
| InChI |
1S/C14H13N3O2/c1-2-19-14(18)17-13-7-10-9-5-3-4-6-11(9)16-12(10)8-15-13/h3-8,16H,2H2,1H3,(H,15,17,18)
|
||||
| InChIKey |
ABHBEIYQTPYXQQ-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Gamma-aminobutyric acid receptor | Target Info | Inhibitor | [1] | |
| Gamma-aminobutyric acid receptor subunit alpha-1 | Target Info | Inhibitor | [1] | ||
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| Retrograde endocannabinoid signaling | |||||
| GABAergic synapse | |||||
| Morphine addiction | |||||
| Nicotine addiction | |||||
| Reactome | Ligand-gated ion channel transport | ||||
| GABA A receptor activation | |||||
| WikiPathways | SIDS Susceptibility Pathways | ||||
| Neurotransmitter Receptor Binding And Downstream Transmission In The Postsynaptic Cell | |||||
| Iron uptake and transport | |||||
| References | |||||
| REF 1 | J Med Chem. 1985 Jun;28(6):824-8.3-Amino-beta-carboline derivatives and the benzodiazepine receptor. Synthesis of a selective antagonist of the sedative action of diazepam. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

